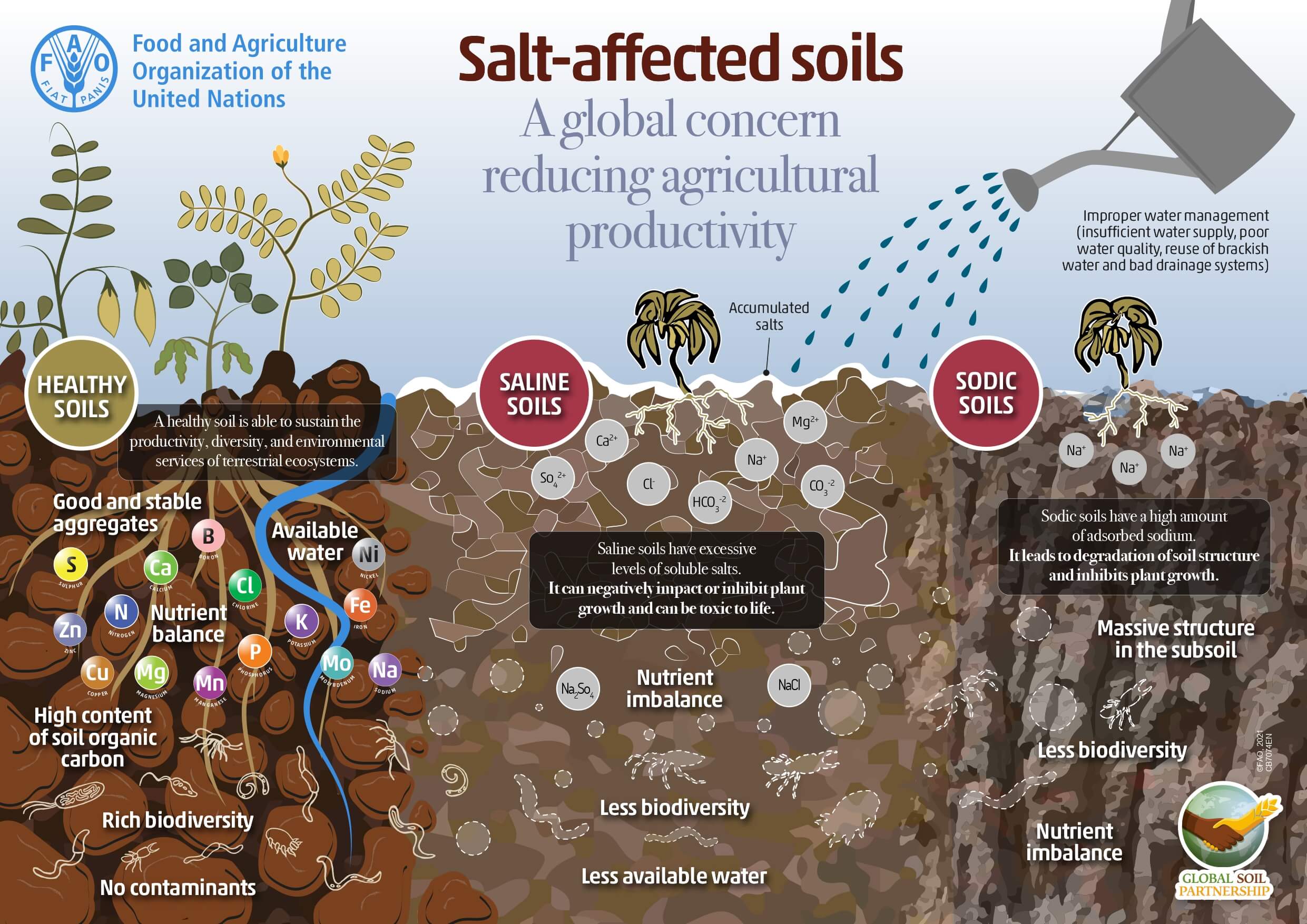
THE PROBLEMS GENERATED BY HIGH SALINITY


- Clogging of the becks, the drops and the pipes of the irrigation system from calcium carbonate deposits.
- Creation of a white crust at the point where the water falls, a phenomenon which appears in the ground especially near the root of the plant.
- Hardening (rock) of the soil because of the concentration of high calcium percentage in the soil = soil permeability.
- Compulsory use of more water.
- Lack of evapotranspiration
- Clogging of nozzles cells as rootstock and capillary vessels of the trunk and the leaves.
- Reduced growth, low productivity and quality depending on the degree of salinity (electrical conductivity Strength).
- Creation of fruit diseases, fungi and algae growth, as growth requires calcium substrate.
- Due to the crystallizing of the ions of calcium carbonate by nature, calcium cannot be absorbed by the plant and this creates adverse conditions for its development

THE CAUSES THAT HIGH SALINITY WATER CREATE PROBLEMS IN IRRIGATION
All natural substances are soluble in water except calcium ions, which when heated and in particular over 65 degrees Celsius is transformed into calcium carbonate. The removal of calcium carbonate deposits can be done with strong acids (eg hydrochloric acid).
Another negative attribute with the calcium crystallization is the fact that they commit all others substances and most importantly nutrient ingredients, which are essential for the plant growth.
Apart from the use of acid and other chemical toxic solution, is the use of electronic devices broadcasting low frequency radio waves in various frequencies.